Families often ask the same core questions: What exactly are we funding? How does this become a real clinical trial? And how do we know safety comes first?
This FAQ was created to walk you step by step through the journey from a drug developed in the lab to a carefully regulated clinical trial in children. We break down complex scientific and FDA processes into plain language, explain why each step is required, and show how your support directly moves the program forward.
Nothing here is rushed or skipped. Every stage is designed to meet FDA standards, prioritize safety, and build the strongest possible case for bringing a promising therapy to our children under proper medical oversight. Our goal is transparency, clarity, and trust so families understand not just what is happening, but why it matters.
Parent Q&A Roadmap: How We Get From Drug to Clinical Trial
1) What is the drug we’re funding?
It’s an mRNA therapy packaged inside a tiny lipid particle that delivers instructions to cells to make a healthy version of the protein our children are missing or not making correctly.
2) What does the lipid particle do?
It acts like a delivery vehicle that protects the mRNA and helps it reach the right cells. The particle is made from components that break down in the body, which is one reason the safety profile has looked promising so far.
3) Is this a real drug or just an idea?
It’s real. The biotech team has already made the therapies for our targets, and they are ready to undergo preclinical testing.
4) Which genes are you focusing on first?
The first focus is on GABRA1 and GABRG2 (and the program can expand). The goal is to deliver healthy protein to help restore function.
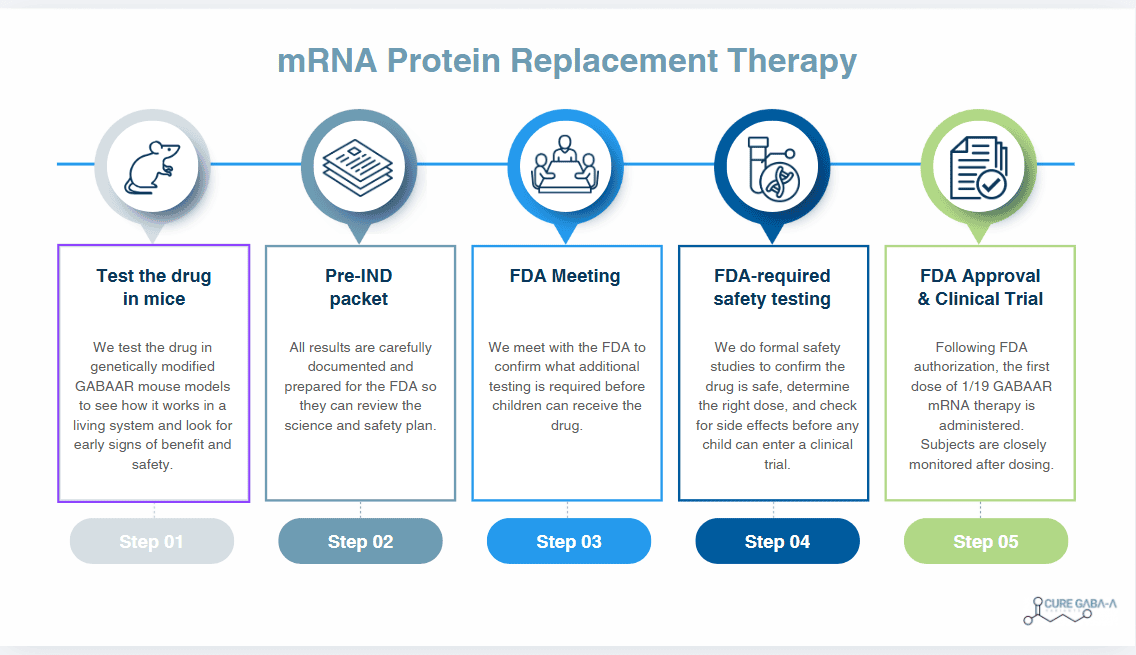
5) Why do you need to test in mice first?
Because the FDA requires evidence that the therapy works in a living system (not just in a lab dish), and is safe enough to move toward human trials. This step creates the “proof” package needed to talk to the FDA.
6) What happens first when the mice arrive?
The mice are checked by veterinary staff and placed in a short quarantine/observation period so they recover from travel and are stable before any research begins.
7) What do researchers measure before treatment starts?
They collect baseline data like weight, behavior, movement, and seizure-related observations (when relevant). This is the “before” picture so we can compare to “after.”
8) What is “efficacy testing” in plain English?
It means: Does the drug actually do what we hope it will do?
Researchers look for signals that the therapy is working in the mouse model over a defined period (often ~6 months of structured testing based on prior rare disease programs).
9) What is “toxicology” and why is it required?
Toxicology is FDA-required safety testing. In plain English, it answers:
- Is it safe?
- What dose is safe?
- Are there side effects or inflammation?
The FDA requires detailed safety data before any child can receive the drug in a trial.
10) What is the “cytokine panel”?
It’s a lab test that checks for signs of inflammation or immune reaction. The biotech described it as a large panel (around 25 markers) that the FDA has accepted in previous programs.
11) What is “biodistribution”?
It means: Where does the therapy go in the body?
Even if the target is the brain, the FDA requires checking multiple organs to confirm there’s no unsafe protein expression elsewhere.
12) What is “pharmacokinetics”?
It means: How long does the therapy stay in the body and how quickly does it break down?
This helps decide how often dosing would be needed in humans.
13) What is a Pre-IND and why does it matter?
A Pre-IND is the first formal submission to the FDA. It shares the plan and early data so the FDA can respond with guidance: keep going, adjust, add tests, or change the plan.
This step prevents delays later.
14) What happens after the Pre-IND?
The team:
- finalizes testing based on FDA feedback
- identifies where the clinical trial will happen (sites/hospitals)
- prepares the full IND submission
15) What is an IND?
IND stands for Investigational New Drug application. It’s the full FDA package (often 1,000+ pages) including manufacturing details, safety results, efficacy data, and trial plans.
When the IND is cleared, children can legally be treated in a clinical trial.
16) What does it cost, in simple terms?
The biotech shared this general estimate:
- Preclinical (animal testing package): roughly $400,000
- Phase 1 clinical trial costs: roughly $1,000,000 (often estimated around $30,000 per patient, plus drug manufacturing and clinical setup)
17) What’s the timeline families should understand?
Based on the biotech’s experience:
- Early efficacy work and Pre-IND preparation can happen in parallel
- Full safety and distribution studies may take about a year
- Trial launch can take additional time to prepare sites and approvals
The overall timeline described aims to move as fast as possible while meeting FDA requirements.
18) What does my donation actually do?
Your donation funds the required steps that unlock FDA permission:
- animal testing to prove safety and function
- the Pre-IND package and FDA meeting
- the IND filing that enables a clinical trial
It turns “a drug in the lab” into “a drug in children, under FDA oversight.”
🔬 CURE GABA-A is a registered 501(c)(3) nonprofit EIN: 93-1989277
Learn more: www.curegabaa.org
💌 Contact us: info@curegabaa.org
📍 Based in the U.S., supporting families worldwide with global branches