The drug exists. The children are waiting. Help us fund the preclinical trial.
“Imagine knowing there’s a drug ready in the lab, the science is solid, and the children who need it are right in front of you, progress is stalled for one reason: lack of funding.
What would you do?”
“Life is genetic Russian roulette, no one knows who will be next. Help us build the cure now, so if it happens to someone you love, the answer will already exist.”
The drug exists. The children are waiting. Help us fund the preclinical trial. That’s the reality for CURE GABA-A families battling GABA-A receptor disorders, a rare and life-threatening category of genetic epilepsies. Some children are trapped in their bodies, bedridden, unable to eat by mouth, and unable to ask for help because they are nonverbal. Others can walk, but are surprised by hundreds of seizures each day. Some seizures are so severe that no medication can stop them, sending families to the ICU again and again. And heartbreakingly, our community has already lost children to this devastating disorder. Help us fund a cure.
“Together, we can go from drug-in-a-fridge to child-in-the-playground.” – Monica Joanna Elnekaveh
Every dollar moves us closer to human trials that could save lives. This isn’t theory. This is ready. The drug is literally sitting in the fridge. We just need your help to take the next step. This is for hundreds of children.
At CURE GABA-A, we’ve made extraordinary progress:
-
- We have funded a targeted mRNA nano lipid particle drug for GABRA1 & GABRG2
- We’ve secured a proof of concept contract with Grann Pharmaceuticals
- We’ve funded and secured the mouse models required to begin testing
- But we are missing one thing: the additional funds to test the target mRNA nana lipid particle drug on the mouse model.
“Imagine knowing there’s a drug ready in the lab, the science is solid, and the children who need it are right in front of you, progress is stalled for one reason: lack of funding. What would you do?” – Agustina Fernandez
mRNA Project: $1.3 Million
Where Your Donation Goes:
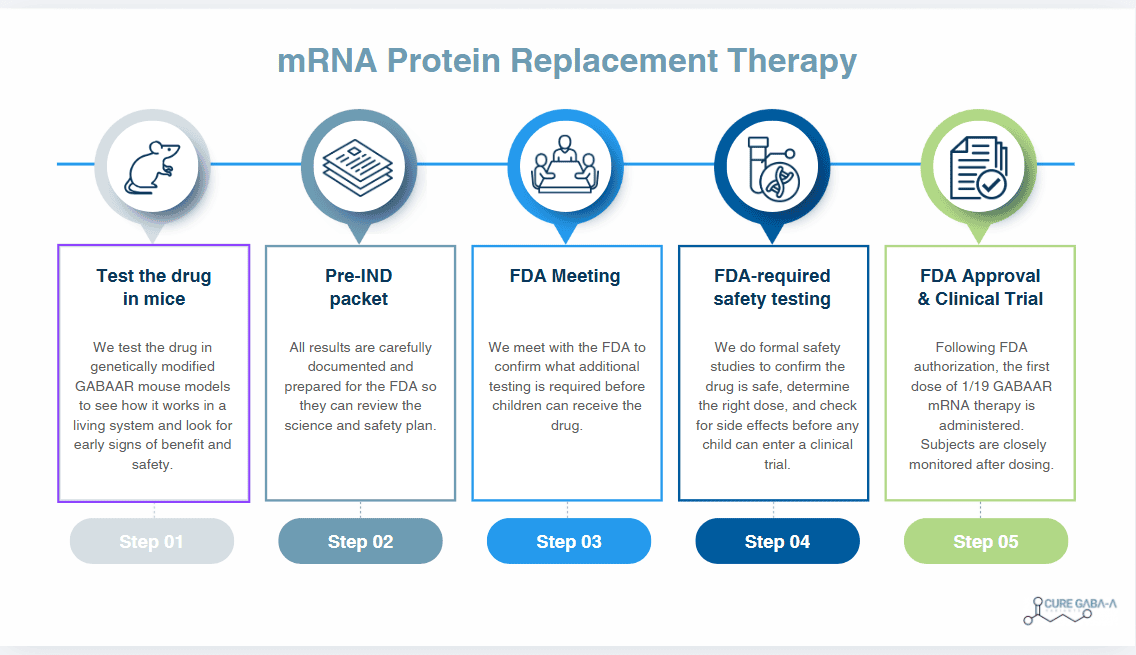
Pre-clinical trial budget is $380,000.
Every donation helps cover:
-
- General lab & cellular work
- Vivarium + mouse testing
- Therapeutic creation costs
- Scientist salaries
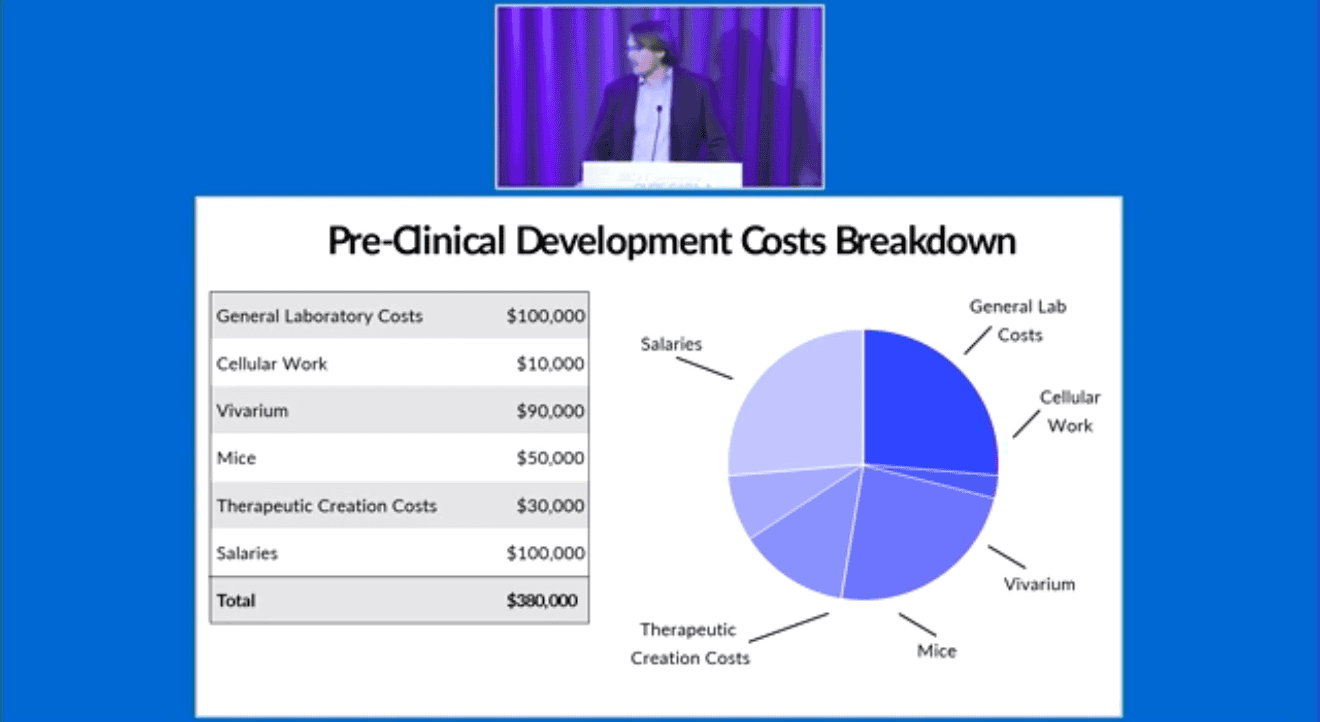
Clinical Development Cost Breakdown $1,000,000:
-
- General FDA expenses
- Clinical Trial Sites
- Patients Costs
- Additional IRB Fees
- Insurance
- Salaries
How You Can Help?
- Donate today — even $10 makes a difference
- Make a tax-deductible gift before year-end
- Ask your employer about company gift matching
- Share this with your community
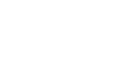
Families often ask the same core questions: What exactly are we funding? How does this become a real clinical trial? And how do we know safety comes first?
This FAQ was created to walk you step by step through the journey from a drug developed in the lab to a carefully regulated clinical trial in children. We break down complex scientific and FDA processes into plain language, explain why each step is required, and show how your support directly moves the program forward.
Nothing here is rushed or skipped. Every stage is designed to meet FDA standards, prioritize safety, and build the strongest possible case for bringing a promising therapy to our children under proper medical oversight. Our goal is transparency, clarity, and trust so families understand not just what is happening, but why it matters.
🔬 CURE GABA-A is a registered 501(c)(3) nonprofit EIN: 93-1989277
Learn more: www.curegabaa.org
💌 Contact us: info@curegabaa.org
📍 Based in the U.S., supporting families worldwide with global branches