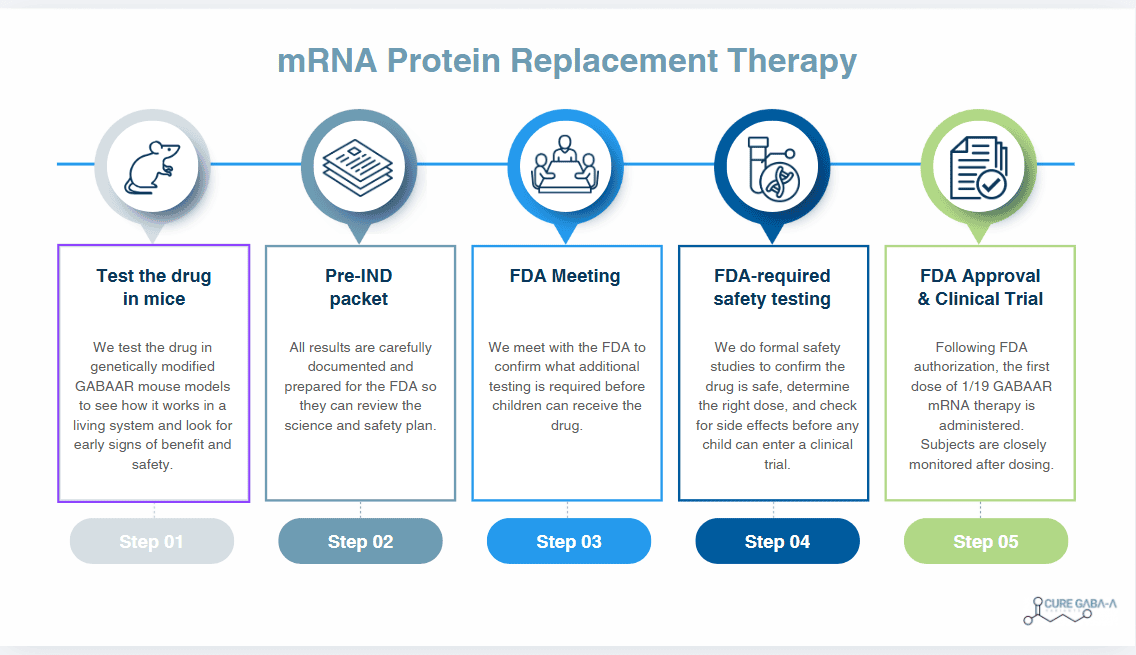
mRNA Protein Replacement: Full Project Overview
This project is designed to move a promising GABAAR mRNA protein replacement therapy from the laboratory into the final stages required before human clinical trials. The work described below represents a carefully planned, FDA-aligned preclinical program that prioritizes safety, scientific integrity, and transparency at every step.
Using validated mouse models developed by leading academic researchers, this study evaluates how the therapy behaves in a living system how it is tolerated, how it functions biologically, and how it can be safely dosed. The data generated here forms the foundation for FDA discussions and is essential for advancing toward clinical trials in children.
This is not exploratory science for curiosity’s sake. It is purpose-driven, regulated research intended to unlock FDA permission to move forward responsibly.
Step 1
Mouse Models Arrive at Grann Pharmaceuticals
Day 0–1
GABRA1 mouse models developed by the Kang Lab at Vanderbilt University arrive at Grann Pharmaceuticals
- Trained veterinary staff perform intake health checks
- Mice are placed in a quiet quarantine and observation area to recover from transport
This ensures the animals are healthy and stable before any research begins.
Step 2
Quarantine & Health Observation
Days 1–3
Mice remain in quarantine for approximately three days
Daily monitoring includes:
- General health
- Eating and activity
- Stress levels following transport
- No drug is administered during this period
Healthy, calm animals are essential for ethical research and reliable data.
Step 3
Baseline Measurements (“Before Treatment”)
Days 3–5
Mice transition from quarantine into the research facility
Researchers collect baseline data, which may include:
- Weight
- Movement and behavior
- Seizure-related observations (when applicable)
All treatment results are measured against these “before” benchmarks.
Step 4
mRNA Treatment Begins
Day 0 of Dosing
- The first dose of the GABRA1 mRNA therapy is administered
- Mice are closely observed following dosing
- Safety and tolerance are the top priority
This marks the first time the therapy is tested in a living system.
Step 5
Repeated Dosing & Continuous Monitoring
Days 0–14 (Every 48 Hours)
mRNA doses are administered every 48 hours
Before and after each dose, researchers assess:
- Overall health
- Behavior
- Any signs of side effects
- Dosing can be paused immediately if concerns arise
This phase establishes safety, consistency, and dosing behavior.
Step 6
Safety & Data Checkpoints
Days 4, 14, and 21
- Planned review points evaluate:
- Safety markers
- Early biological signals
- Study progression and integrity
These checkpoints are required to build a strong FDA pre-IND data package.
Step 7
Long-Term Follow-Up & Monitoring
Day 22 through Day 175
Mice continue to be monitored after dosing ends. Follow-up assessments occur approximately every 28 days.
Researchers evaluate:
- Longer-term data strengthens the case for FDA discussions and human trials.
- How This Leads to FDA Approval Discussions
All data from this study contributes directly to:
- Safety evidence
- Proof of biological activity
- Dose planning
- FDA pre-IND discussions for GABRA1 or G2 or B3
This study represents one of the final steps before asking the FDA for permission to begin human clinical trials.
🔬 CURE GABA-A is a registered 501(c)(3) nonprofit EIN: 93-1989277
Learn more: www.curegabaa.org
💌 Contact us: info@curegabaa.org
📍 Based in the U.S., supporting families worldwide with global branches